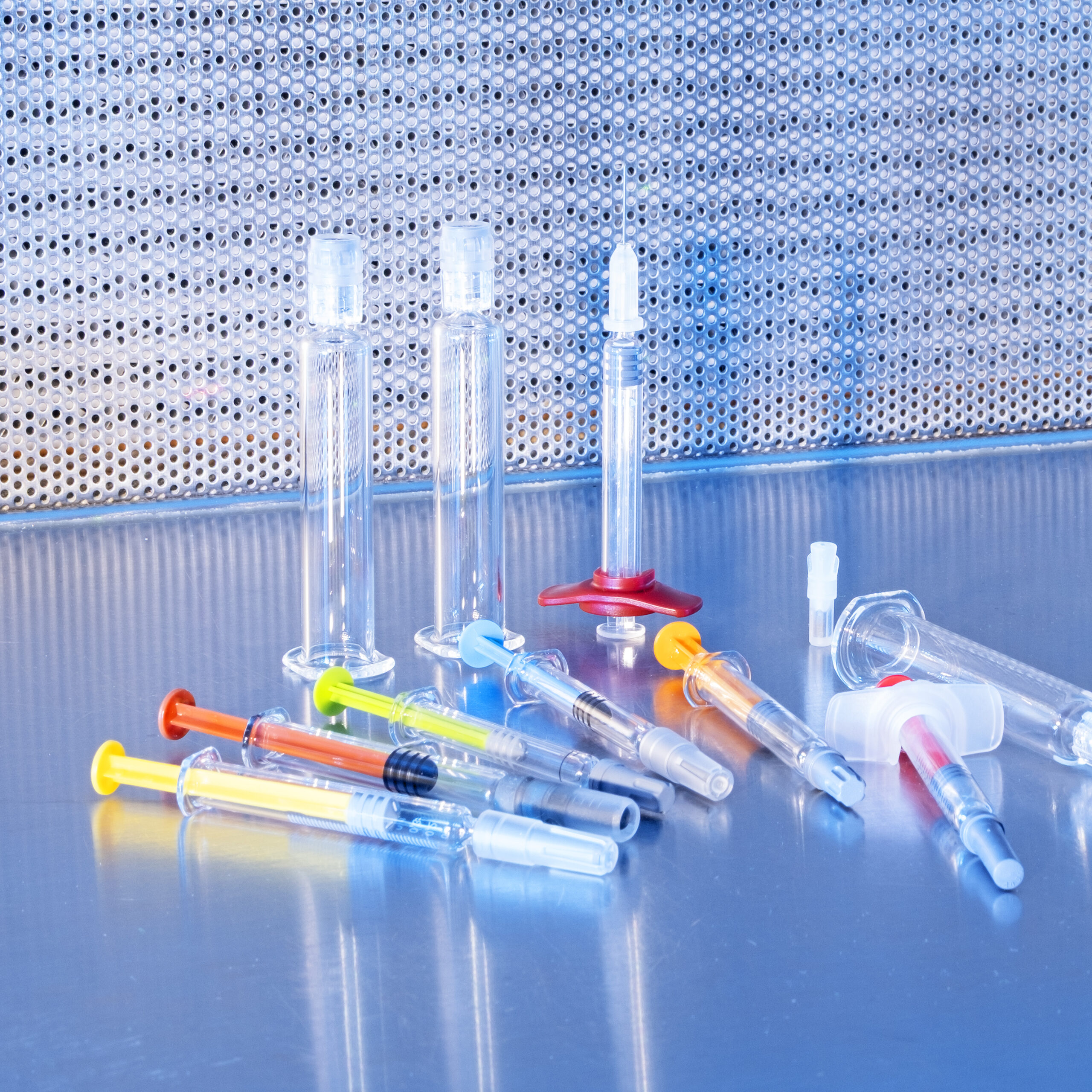
Syncuris GmbH – Welcome to Your small-scale Fill & Finish CDMO
OUR MISSION
The answer to an industry bottle neck in pharmaceutical development:
Agile, state‑of‑the‑art filling and aseptic assemblyservices that enable biotech and pharmaceutical companies to bring safe, effective and convenient injectable therapies to patients swiftly and reliably.
OUR SERVICES
We provide end-to-end support for pharmaceutical development, filling and supply chain needs, starting with formulation strategy & process consulting, including packaging development and clinical supply services.
We deliver state-of-the-art support for biotech and pharma companies with the need for small and flexible filling capacities. We currently offer services for technical batches and will expand into GMP/Annex 1 compliant services soon.
Todays biologics and other injectables often require delivery devices such as injectors, pens, inhalers or more. Syncuris will offer aseptic assembly of filled primary containers with those devices.
With our services, you bring new, effective, safe and convenient therapies to your patients – fast and reliable!

Project Management & Consulting Services
- Sourcing of APIs, Excipients & Packaging Materials
- Bulk Filling & Technical Batches
- GMP-Compliant Documentation
- Project Management & Regulatory Affairs

Fill & Finish for Technical Batches
- Formulation Development
- Process Development
- Filling of technical batches
- ICH Stability studies
Life Science Expertise
at your Finger Tips

Dr. Stefan Henke
Managing Director Operations
Dr. Stefan Henke is a senior pharmaceutical executive and Qualified Person with extensive leadership experience in R&D, GMP operations, and innovative drug delivery technologies. He has led major biotech and OTC development organizations, built GMP facilities, managed international partnerships, e.g. at IIS Gmbh, LTS Lohmann Therapie-Systeme, Merz Consumer Care GmbH, Merck Selbstmedikation GmbH, Boehringer Ingelheim Vetmedica GmbH and Schering AG.

Axel Fischer
Managing Director Business Development
Axel is an experienced entrepreneur and leadership expert with 30 years of experience in the life sciences industry. As founder and CEO of Activoris, he has built a group of several business units in the fields of medical technology, pharmaceuticals, and food.
Axel held several management and board positions in medtech and pharma companies and is a serial entrepreneur, e.g. Activoris, Actarmo Medical, SilviMed. He focuses on scaling start-ups, technology transfer, strategic deveopment and collaborations.

Dipl.-Ing. Klaus List
Process Development, Qualifications
Klaus is the owner and Managing Director of IBL GmbH. With over 30 years of experience in product and process development within GMP-regulated pharmaceutical and medical device manufacturing, he has worked with leading companies such as Bayer, Boehringer Ingelheim, and Roche. He now contributes this expertise as a plant engineer at Syncuris GmbH

Dr. Stefan Degenhardt
CFO
Stefan has more than 25 years of experience in senior finance and management in the life science industry. He is co-founder of Activoris Medizintechnik GmbH and also serves as managing director of AspiAir GmbH, a Biotech company focusing on the development of non-opioid treatments for pain and migraine attacks.
Stefan held management and finance positions at various pharmaceutical and life science companies. In his position as CFO of Activaero, he played a key role during the € 130m exit to Vectura plc.
News & Events
Latest News
- Feb. 26, 2026: New web site launched
Meet Syncuris at the following Events:
- Stefan Henke will attend the 15th World Meeting on Pharmaceutics, Biopharmaceutics and Pharmaceutical Technology, March 23-26 in Prague. Arrange a meeting >>


Syncuris GmbH
Wohraer Str. 37
35285 Gemünden (Wohra)
GERMANY
Get in touch
Mail: info@syncuris.com
Imprint & Disclaimer
Cookies